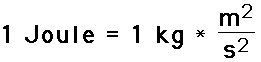Heat Transfer: Heat transfer is a discipline of thermal engineering that concerns the generation, use, conversion, and exchange of thermal energy and heat between physical systems
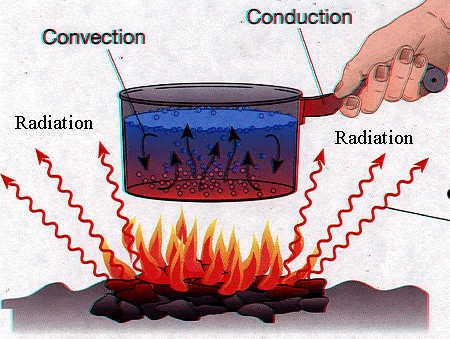
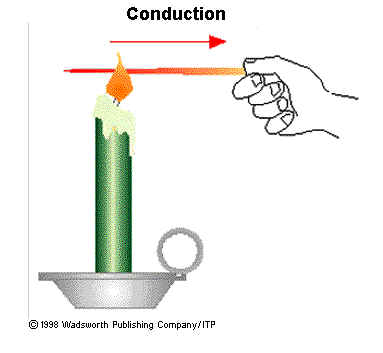
Conduction: the process by which heat or electricity is directly transmitted through a substance when there is a difference of temperature or of electrical potential between adjoining regions, without movement of the material.
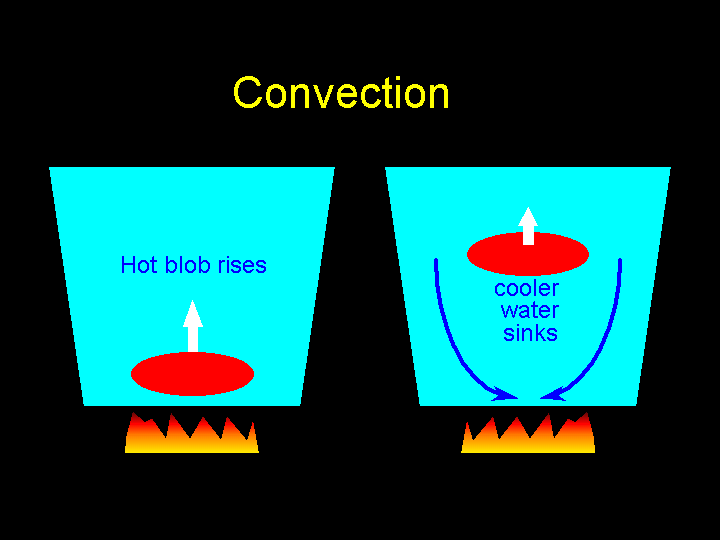
Convection: the movement caused within a fluid by the tendency of hotter and therefore less dense material to rise, and colder, denser material to sink under the influence of gravity, which consequently results in transfer of heat.
Radiation: the emission of energy as electromagnetic waves or as moving subatomic particles, esp. high-energy particles that cause ionization.
Joule: the SI unit of work or energy, equal to the work done by a force of one newton when its point of application moves one meter in the direction of action of the force, equivalent to one 3600th of a watt-hour
Calories: _either of two units of heat energy.the energy needed to raise the temperature of 1 gram of water through 1 °C (now usually defined as 4.1868 joules).
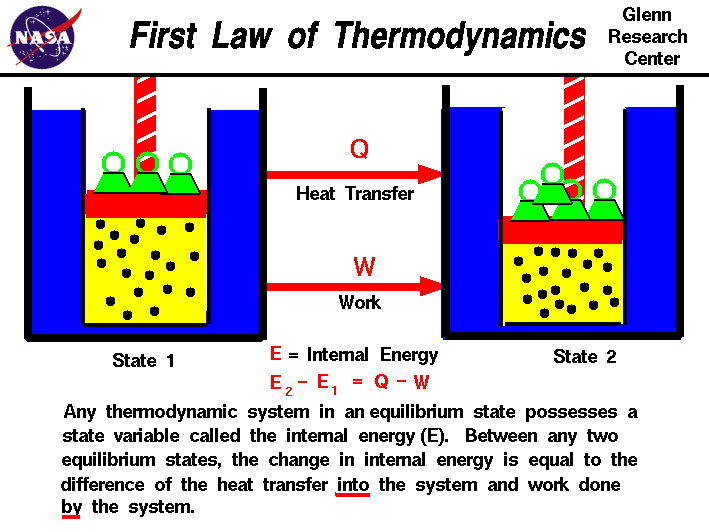
1st Law of Thermodynamics: The first law makes use of the key concepts of internal energy, heat, and system work. It is used extensively in the discussion of heat engines.