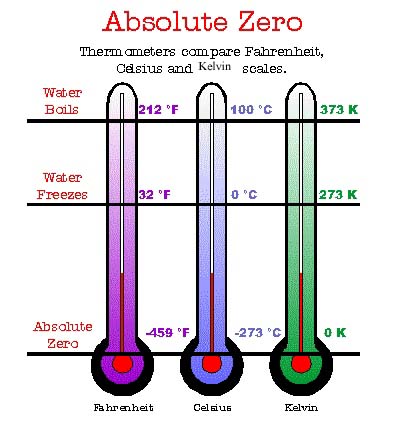
* Absolute zero : The temperature at which a substance has no kinetic energy per particles
to give up. This temperature corresponds to 0 K, or to - 273*C.
to give up. This temperature corresponds to 0 K, or to - 273*C.
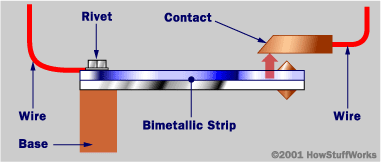
* Bimetallic strip: two strips of different metals, such as one of brass and one of iron, welded or riveted together into one strips .because the two substances expand at different rates, when heats or cooled the strip bends. Used in thermostats.
*Calorie: A unit of heat. One calorie( symbol cal) is the heat required to raise the temperature of one gram of water one Celsius degree. one Calories( with a capital C) us equal to one thousand calories and it unit used in describing the energy available from food. 1 calorie =4.18400 joules.
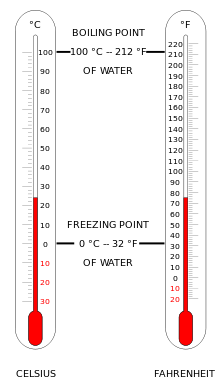
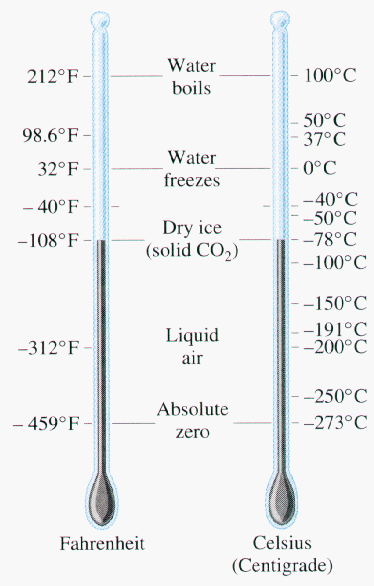
*Celsius scale: A temperature scale with 0 as the melt-freeze temperature for water and 100 as the boil condense temperature of water at standard pressure( one atmosphere at sea level.
*Fahrenheit Scale: The temperature scale in common use in the United States. number 32 is assigned to the freezing point of water and number 212 to the boiling point of water.
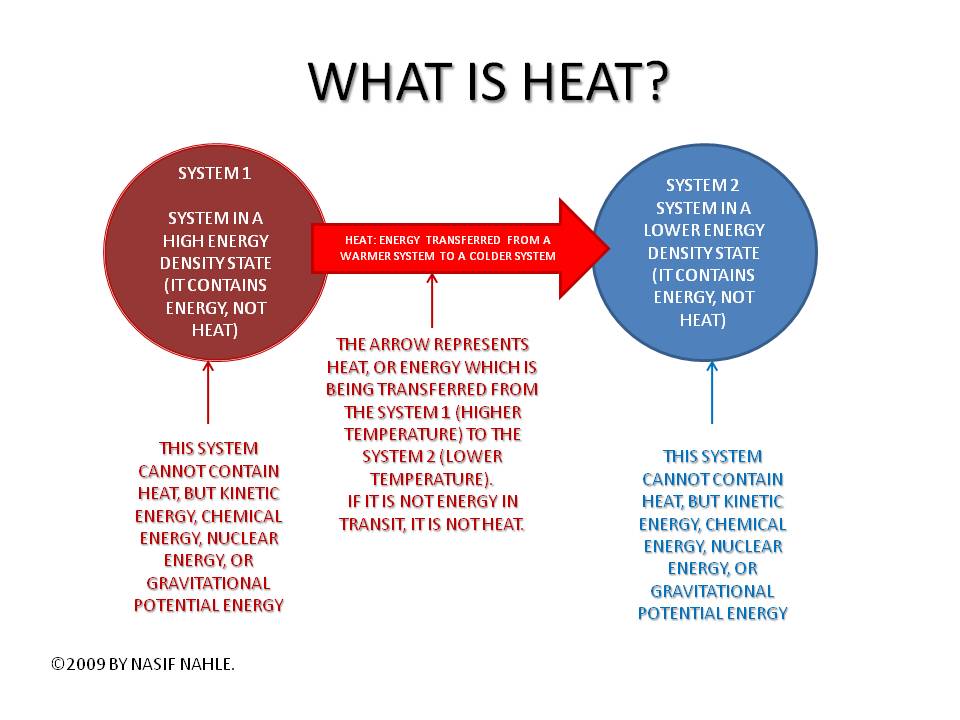
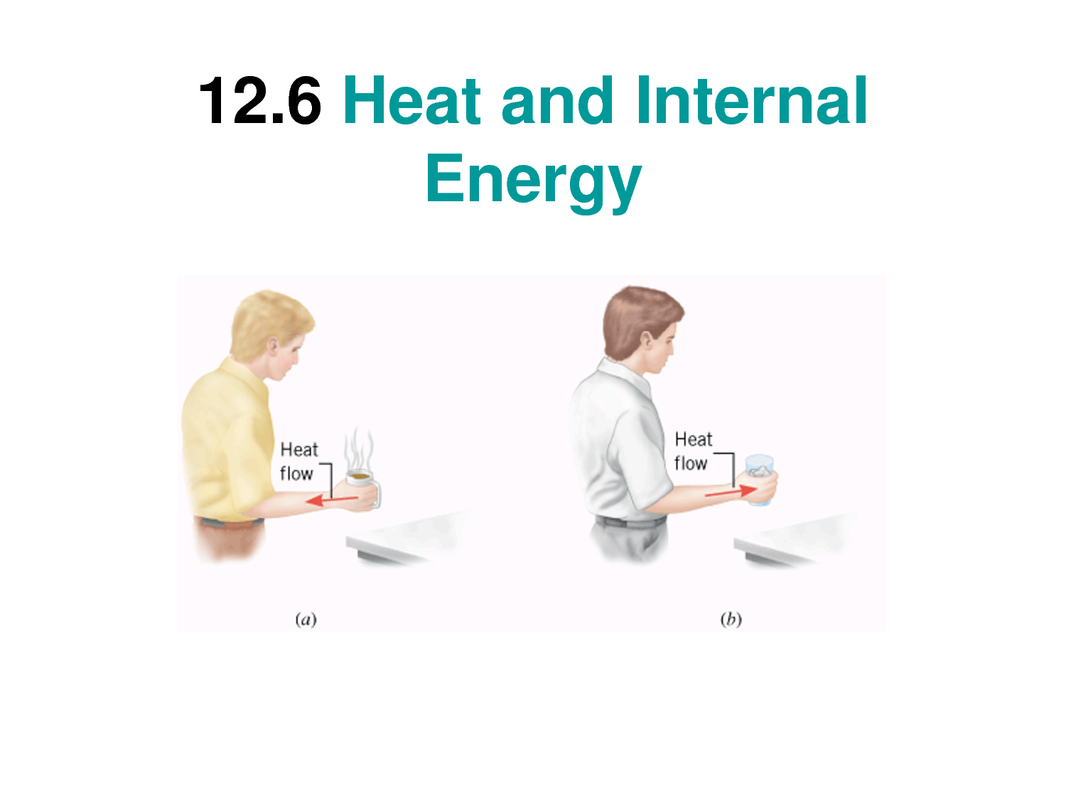
*Heat: Energy transfer via random molecular motions, resulting in gain or loss of internal energy.
*Internal Energy: The total energy stored in the atoms and molecules within substance.
*Kelvin Scale: a scale of temperature with absolute zero as zero, and the triple point of water as exactly 273.16 degrees.
*Kilocalorie: a unit of energy of 1,000 calories (equal to 1 large calorie).
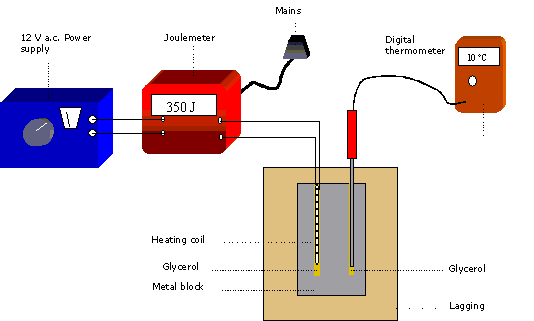
*Specific heat capacity: the heat required to raise the temperature of the unit mass of a given substance by a given amount (usually one degree).
*Temperature: the degree or intensity of heat present in a substance or object, esp. as expressed according to a comparative scale and shown by a
thermometer or perceived by touch.
thermometer or perceived by touch.
*Thermal contact: the state of two or more object or substances in contact such that it is possible for heat to flow from one object or substance to another.
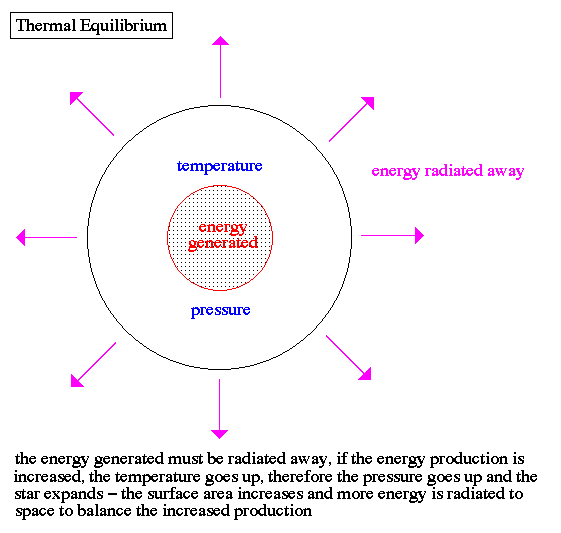
Thermal equilibrium: the state of two or more object or substances in thermal contact when they have reached a common temperature.
*Thermostat: a type of valve or switch that responds to changes in temperature and that is used to control the temperature of something.